Initial Symptoms and Misdiagnosis
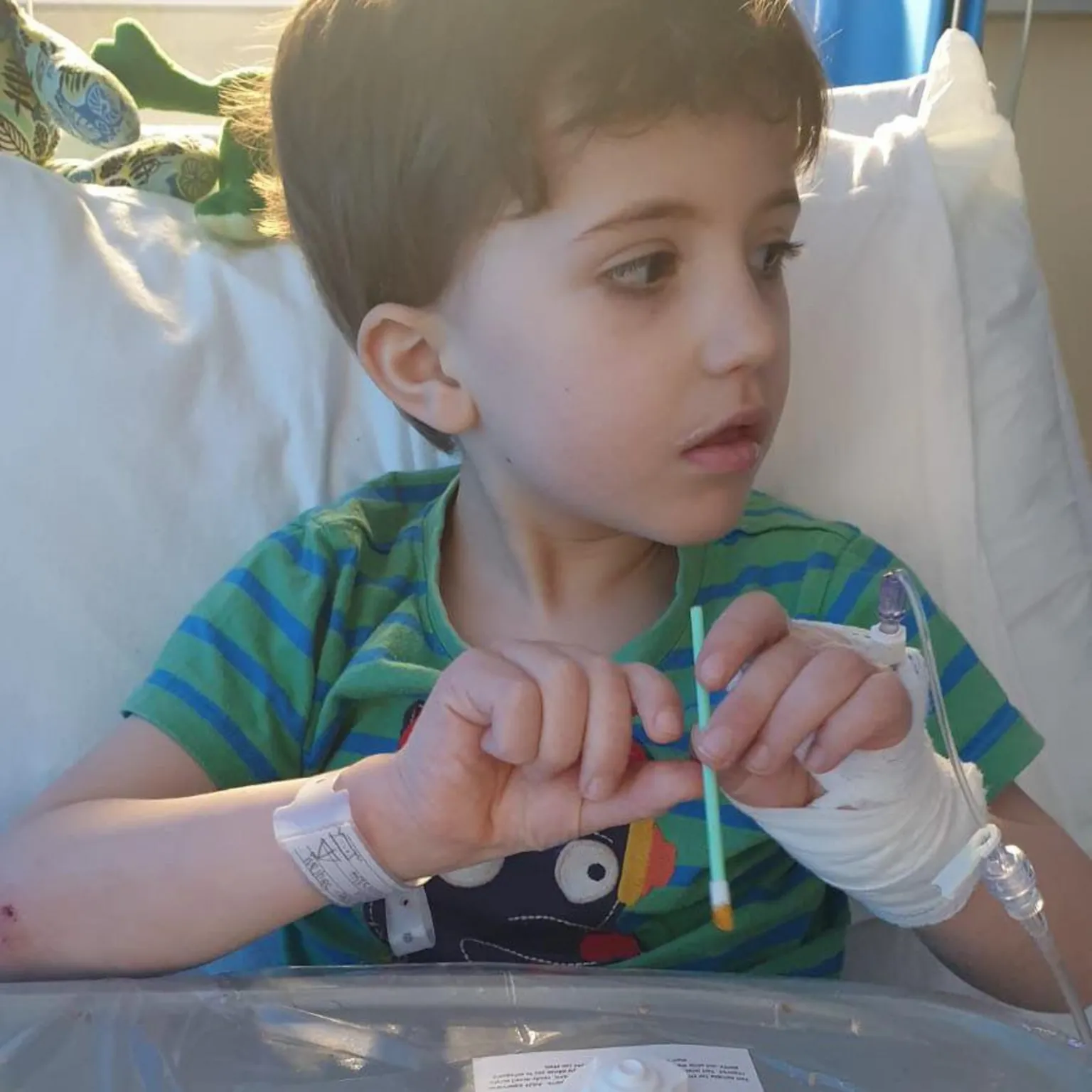
When seven-year-old Roo suddenly became unwell last year, experiencing weight loss and excessive thirst, his doctors and parents feared he might have a brain tumour.
However, medical investigations revealed that Roo had been accidentally poisoned by an overdose of vitamin D, which had been prescribed to alleviate his growing pains.
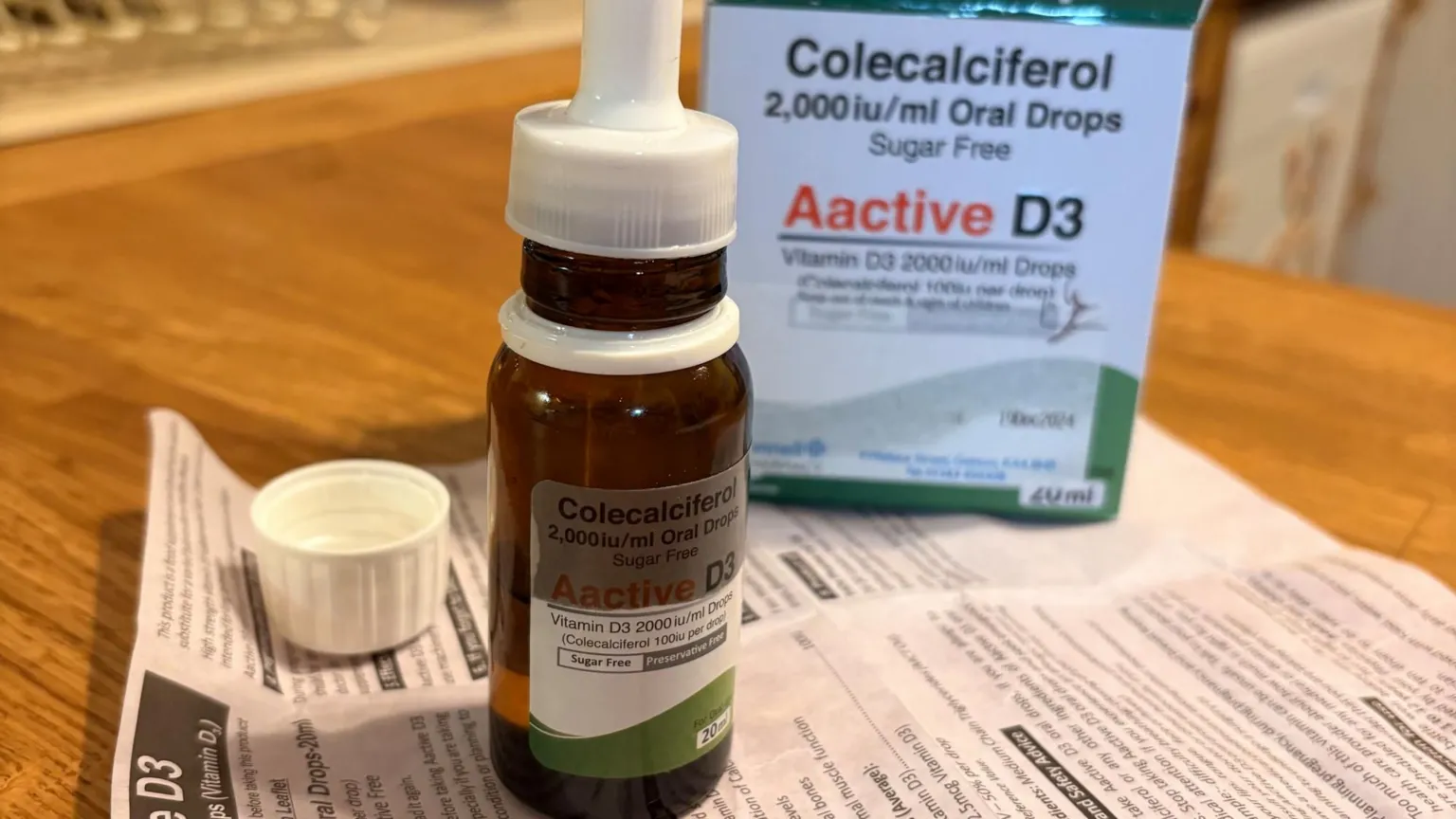
Roo's bottle of vitamin D3 drops was found to be approximately seven times more concentrated than it should have been. This bottle was part of one of two defective batches distributed across the UK.
The excessive dosage caused Roo to suffer an acute kidney injury. A leading expert informed that the young child would have died had he completed the prescribed course.
Vitamin D is an essential nutrient that regulates calcium and phosphate levels necessary for healthy bones, teeth, and muscles. It is commonly taken as an over-the-counter supplement by millions of adults.
However, higher-dose vitamin D, prescribed by doctors, is still classified as a food supplement rather than a medicine and is therefore not regulated by the Medicines and Healthcare products Regulatory Agency (MHRA).
Instead, vitamins and food supplements fall under the oversight of the Food Standards Agency (FSA).
The MHRA stated that it collaborates with the FSA to ensure public safety.
A leading expert has suggested to that the MHRA should reconsider the regulatory framework for vitamin supplements.
Roo was prescribed a high dose of vitamin D3 drops for 12 weeks starting in December 2024 to relieve severe leg pain.
This followed a referral to paediatricians at Crosshouse Hospital near his home in Kilmarnock, Ayrshire.
Blood tests indicated Roo was generally healthy except for a slightly low vitamin D level.
He was given vitamin D3 supplements—also known as colecalciferol—to increase his blood levels.
Over the following weeks, Roo became lethargic and lost his appetite.
He began to lose weight and drank water excessively, described by his mother, Carys Hobbs-Sargeant, as "like he was in a desert."
Roo also experienced vomiting throughout January and had difficulty eating.
"He was really sick," Carys said.
"He lost more than 10% of his body weight over that six weeks. He had these huge baggy eyes and he was so tired and he just couldn't eat."
Upon a follow-up with the paediatrician, Roo was immediately admitted to hospital.
Initial blood tests revealed an acute kidney injury and severe dehydration of his kidneys, prompting doctors to investigate the cause.
Initially, there was no concern about the vitamin D prescription, as Roo was only two-thirds through the recommended course.
However, his condition worsened.
"He became hypercalcaemic, and they were very concerned that his calcium was so high in his blood," his mother said.
"They were looking at whether it was a brain tumour, and we were sort of gearing up for him to have an MRI of his brain."
Roo's case was also reviewed by teams at Glasgow's Royal Hospital for Children. A chance phone call with an endocrinologist there provided a critical clue.
A colleague in Manchester inquired whether they had encountered any cases involving a "bad batch" of vitamin D3.
With the batch details, Roo's medical team cross-referenced the bottle he was using daily.
"We flipped from it was his body doing something weird to he's essentially been poisoned by this bad batch," said Carys.
"You felt relieved and lucky and angry all at the same time that it wasn't cancer, but it was something that had happened to him. It's poisoning, essentially."
Roo's bottle was linked to one of two faulty batches of Active D3 supplements from TriOn Pharma, distributed throughout the UK.
The drops contained seven times the intended concentration.
The faulty batches were recalled by the Food Standards Agency on 9 January, but Food Standards Scotland (FSS), responsible for the recall in Scotland, reported that the alert did not reach the appropriate departments.
An investigation found that the Ayrshire pharmacy dispensing the drops did not see the manufacturer's recall email until nearly three months after it was issued.
MHRA medicine alerts would have been sent to the pharmacies' high-priority clinical inboxes and should have been read immediately.
Prof Stuart Ralston of the University of Edinburgh, former chair of the MHRA's Commission on Human Medicines, stated that Roo's case highlights the need for MHRA to consider regulating vitamin supplements.
"The product was estimated to have seven times the amount of vitamin D that he should have had," he said.
"I have not come across that in a career of over 40 years.
He was meant to have a 12-week course. If he'd done that it would be 3.5 million units and luckily he did not, because if he had, he'd have died undoubtedly."
Ralston added that if the supplement had been regulated as a medicine, Roo's GP would likely have received an urgent alert.
The MHRA reiterated that dietary supplement regulation falls under the FSA's remit and that both agencies cooperate to protect the public.
FSS stated it works closely with the FSA and local authorities to ensure effective communication of product recalls.
The Scottish government informed Carys that it had raised concerns about regulation and supplement recalls with the UK Department of Health and Social Care.
Similar Case: Kayan Khan
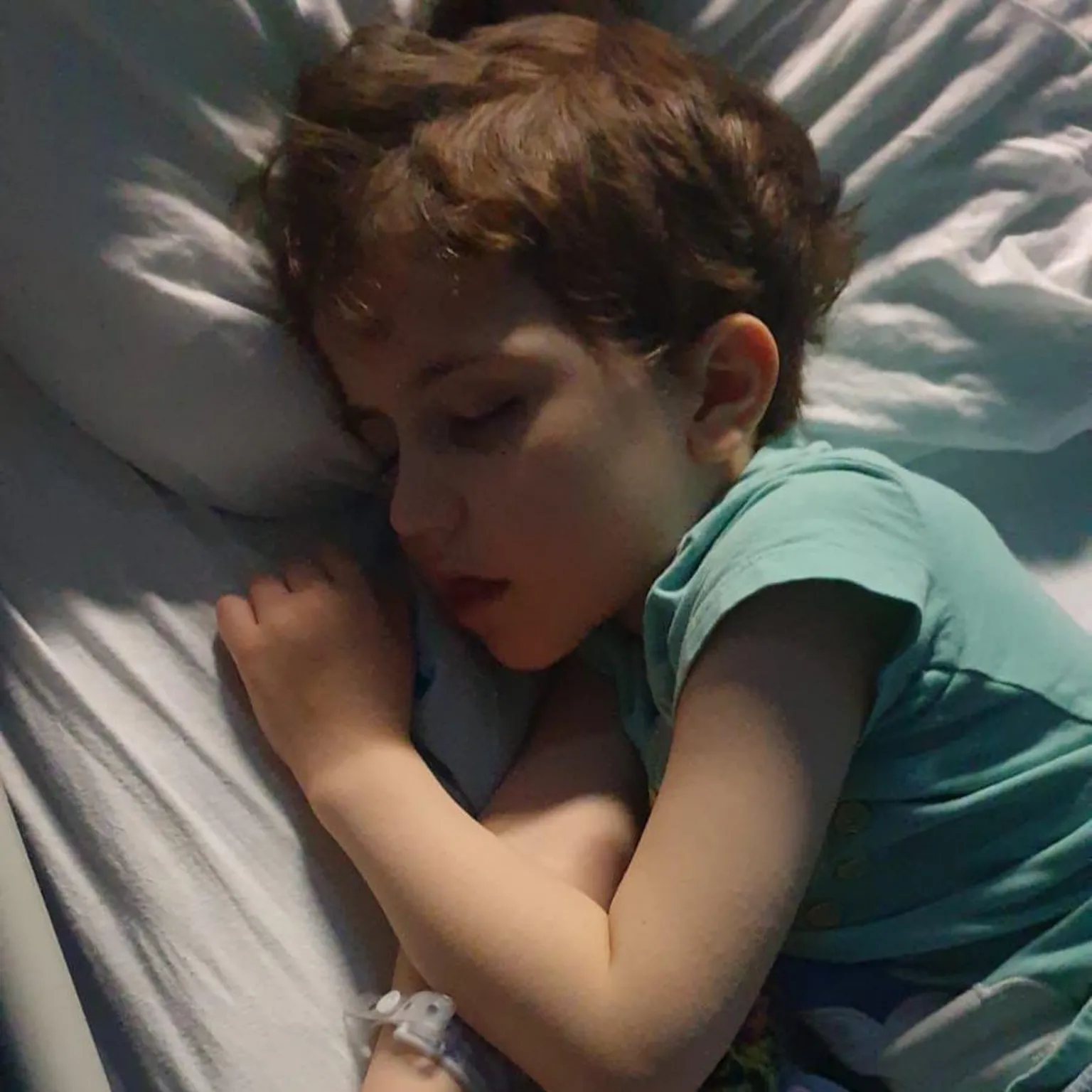
A month before Roo began his vitamin D3 course, Kayan Khan from Sheffield was prescribed the same brand of supplements.
Kayan, now 13, has multiple complex medical conditions, including chronic kidney disease. His GP prescribed an eight-week course of vitamin D consistent with recommendations.
In February 2025, Kayan was admitted to Sheffield hospital with elevated calcium levels and impaired kidney function.
A letter to his mother, Alaina, from Sheffield Children's NHS Foundation Trust indicated that doctors struggled to control his calcium levels and diagnosed vitamin D intoxication "far above expectation for standard supplementation."
Alaina had not retained the bottle of vitamin drops, so the batch could not be confirmed, but the letter noted that the "logical explanation" for his toxicity was increased vitamin D intake.
Again, news of the FSA recall had not reached Alaina, although hospital doctors and local pharmacies had been informed.
She only learned about the recall in April 2025.
Kayan experienced significant deterioration in kidney function. Although there has been some recovery, he may require a transplant in the future.
"As a parent, as a mum, I've been let down," she said.
"I can't help but feel that for eight weeks, I'd been slowly poisoning my son."
Dr Jeff Perring, executive medical director at Sheffield Children's NHS Foundation Trust, explained that while GPs prescribe medications, the hospital follows strict procedures to inform patients and families about recalls for medications prescribed directly.

Manufacturer and Regulatory Responses
TriOn Pharma, manufacturer of the drops, stated they initiated a prompt recall and notified the FSA and MHRA as required after identifying the issue.
"Recall notifications were issued to all direct customers through established channels for onward communication across the supply chain," they said.
"We continue to work with authorities to support patient safety."
Long-Term Impact on Roo
Roo's vitamin D levels took approximately a year to return to a safe range.
Carys indicated that the long-term effects remain uncertain.
"He went from an outdoor, forest school kid to someone who now needs a wheelchair to go longer distances," she said.
"He hasn't immediately gone back to who he was before and he's still dealing with that on a day to day."


Vitamin D Supplementation and Regulation
Vitamin D is essential for maintaining calcium and phosphate balance, supporting healthy bones, teeth, and muscles. While low-dose vitamin D supplements are widely available over the counter, higher-dose preparations prescribed by doctors remain classified as food supplements rather than medicines.
This classification means they are regulated by the Food Standards Agency rather than the MHRA, which oversees medicines.
The case of Roo and Kayan has raised questions about this regulatory approach, with experts suggesting that vitamin supplements, especially at higher doses, should be subject to stricter oversight to prevent similar incidents.
Summary
The accidental poisoning of Roo with a highly concentrated vitamin D supplement highlights significant gaps in the regulation and communication surrounding vitamin supplements in the UK. Delayed recall notifications and classification of high-dose vitamin D as a food supplement contributed to serious health consequences for Roo and Kayan Khan. Experts urge a review of regulatory frameworks to enhance patient safety.